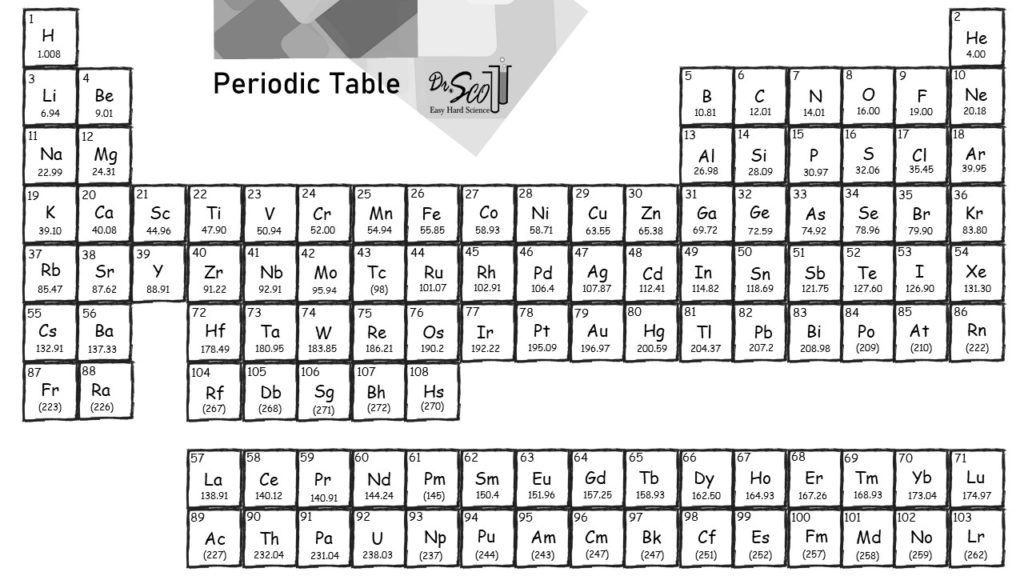
The percentage of a commodity which is recycled. The number of atoms of the element per 1 million atoms of the Earth’s crust. This is calculated by combining the scores for crustal abundance, reserve distribution, production concentration, substitutability, recycling rate and political stability scores. The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. Metallic character decreases from left to right in each period of the periodic table.A vertical column in the periodic table. 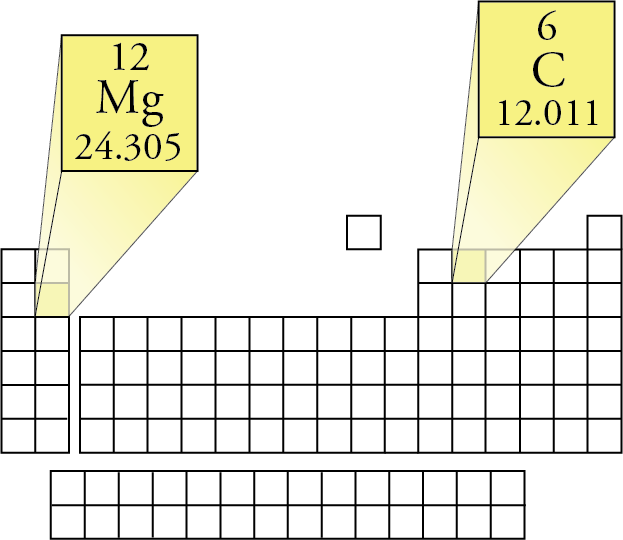
Metallic character increases down a group. Going down a group, the atomic radius increases: the outer electrons are farther from the nucleus and are easier to remove, thus increasing the metallic character.The metallic character is essentially the ease with which an atom gives up an electron :max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
Metalloid: an element with properties intermediate between metals and nonmetals. Metalloids divide the periodic table in a zigzag line between metals on the left and nonmetals on the right.Nonmetal: poor conductor of heat and electricity. They are generally softer elements, often colored, with lower melting and boiling points than most metals.Metal: good conductor of heat and electricity. They tend to be hard, metallic-looking solids with high melting and boiling points. The majority of elements are metals.Most elements can be classified as metals or nonmetals based on their ability to conduct heat and electricity: ⇒ very stable elements, generally considered to be inert gases Noble gases: last column = helium, neon, argon, (krypton, xenon, radon).⇒ colorful and corrosive nonmetals, very reactive Halogens: penultimate column = fluorine, chlorine, bromine, iodine, (astatine).⇒ react with O 2, less reactive to water than alkali metals Alkaline-earth metals: 2 nd column = beryllium, magnesium, calcium, (strontium, barium, radium).⇒ very reactive metals and can explode if they are exposed to water Alkali metals: 1 st column = lithium, sodium, potassium, (rubidium, caesium and francium).Several groups of representative elements are known by common names: The top-row elements are called lanthanides and the bottom-row elements are actinides The inner transition metals: elements in the 2 rows at the bottom of the table.The transition metals: elements in the groups between the elements of the main group (groups 3-12).The main-group elements: elements in the 2 groups on the far left and the 6 groups on the far right of the periodic table (groups 1, 2, 13-18).Members of the same group have the same number of electrons in the outermost shells of their atoms and form bonds of the same type. Here He Lies Beneath Bed Clothes, Nothing On, Feeling Nervous, Naughty Mar gret Always Sighs, " Please Stop Clowning Around "Ī vertical column of elements is called a group. 3 rd period: Na, Mg, Al, Si, P, S, Cl, Ar.You should generally know the first 3 periods: A period forms a row of the periodic table. 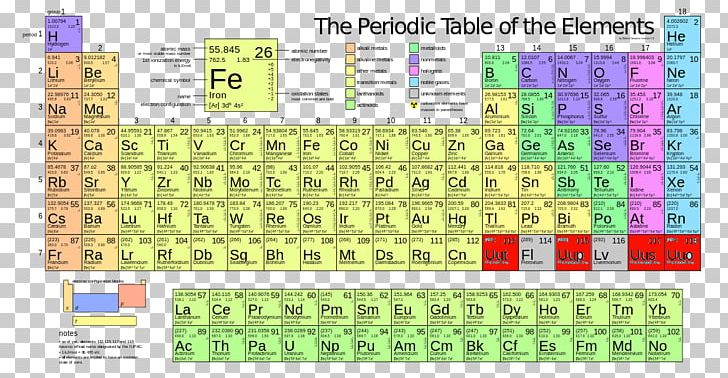
The chemical elements have a periodic pattern when they are arranged in ascending order of atomic number. Each vertical column corresponds to a group of elements with the same properties, each horizontal line corresponds to a periodĮach element of the periodic table is represented by a tile containing the name of the element (chemical symbol composed of one or two letters), the atomic number and the average mass (usually indicated with 4 significant figures) The elements are arranged by atomic number. A chart of 118 elements first published by Dmitri Mendeleev in 1869, in which elements with similar chemical and physical properties are grouped.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
